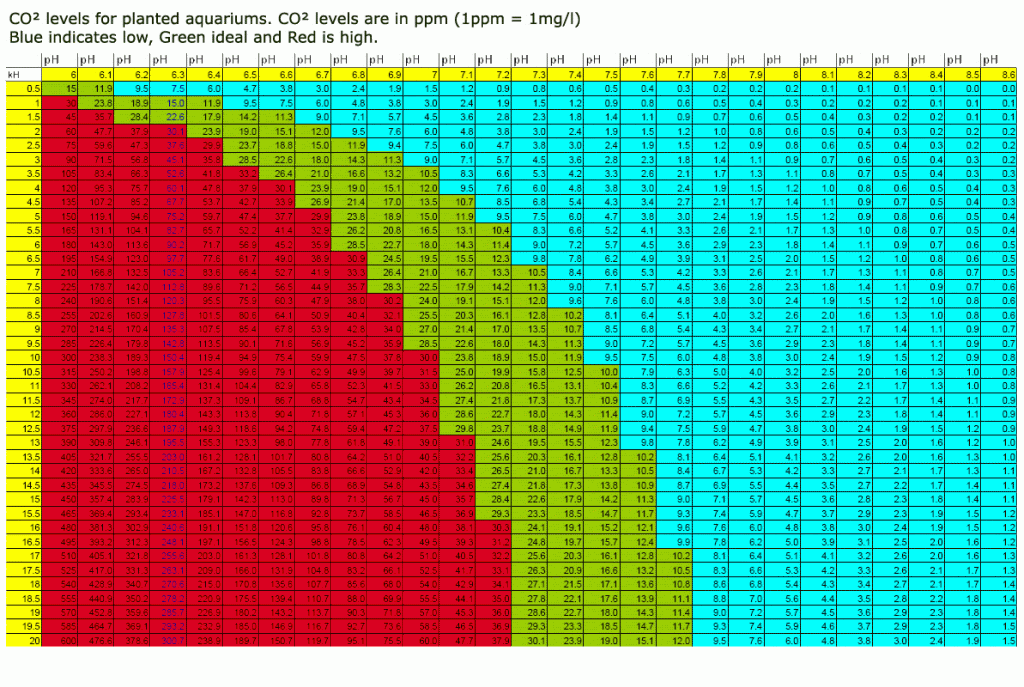
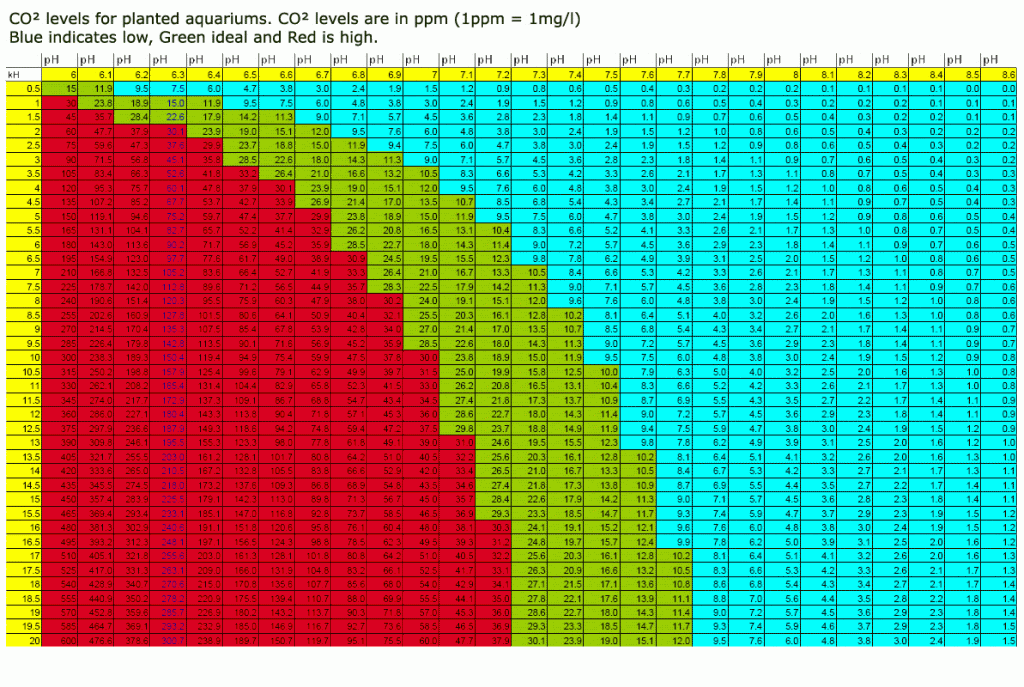
Yes, you're correct about the chart, however the chart is only accurate if the KH is entirely carbonate, which might not be the case. It should be mostly carbonates, however at higher KH readings especially, there could be a few degrees of non-carbonate KH that will throw off the calculation, likely by as much as 10 ppm or greater. (This is why a reference solution is used for drop checkers.)
By off-gassing a sample of tank water, the co2 concentration will be at equilibrium with the surrounding air, and we can assume that this concentration is roughly 3 ppm of co2. If you look for a 3.0 ppm value anywhere on the chart, and follow the same row to the left to reach a pH drop of 1 full degree, you'll see a value of 30 ppm. I hope this helps.